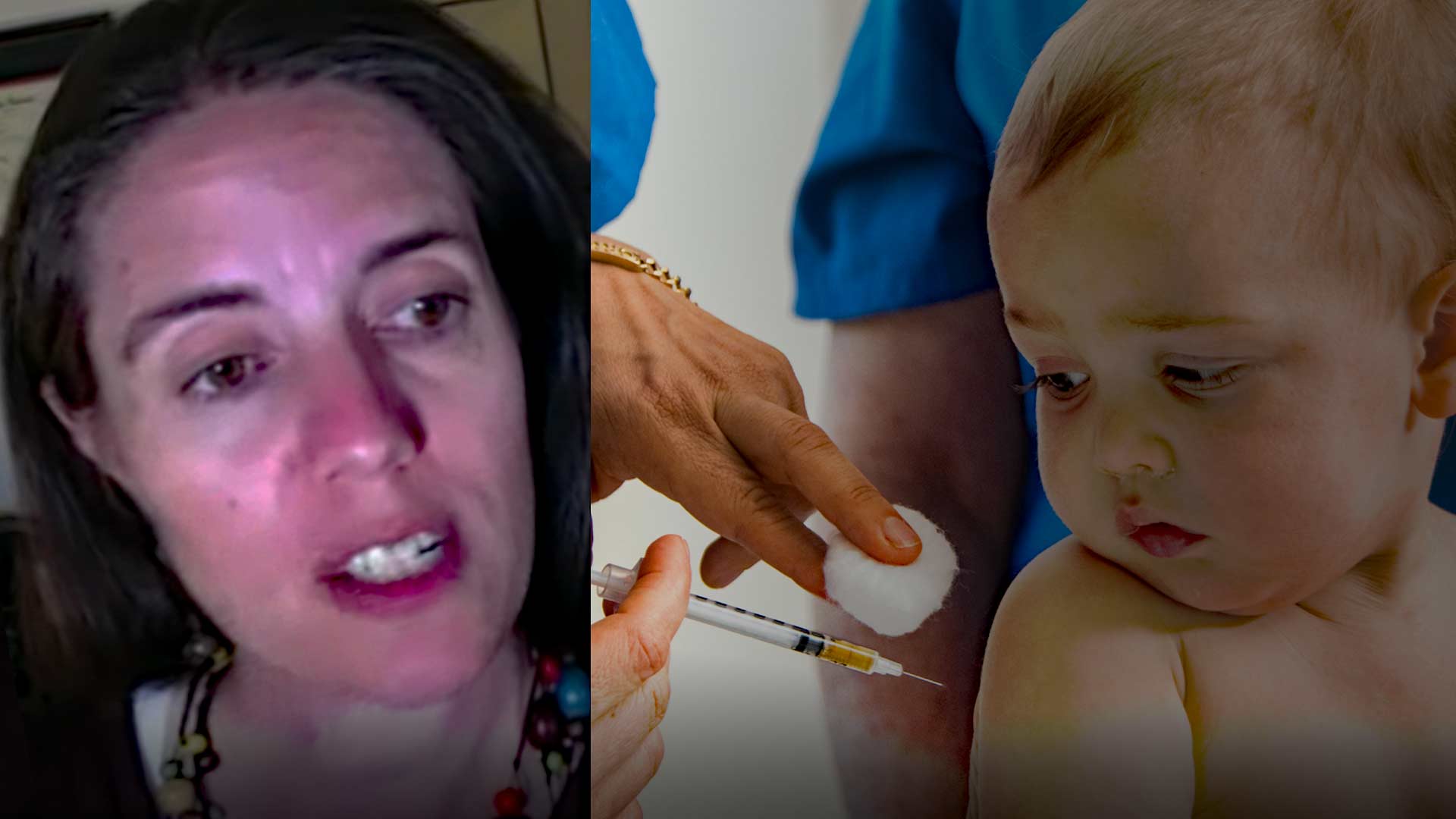
Led by Alaska’s Chief Medical Officer Anne Zink, the powerful Association of State and Territorial Health Officials (ASTHO) is urging “all Americans over six months” get yet another Covid jab update.
In addition to being appointed by Gov. Mike Dunleavy to spearhead Alaska’s Covid response, Zink serves as president of ASTHO, a wealthy and influential nonprofit that includes more than 100,000 public health professionals across the nation. The high-profile position gives her a national voice when it comes to U.S. health policy.
On Sept. 15, her organization issued a complete endorsement of the FDA’s controversial recommendation that every American older than six months get an updated Covid shot. The endorsement included a statement by ASTHO’s Chief Medical Officer Marcus Plescia, who claimed that vaccination is “one of the most effective ways to keep people safe from COVID-19 by preventing serious illness, hospitalization, and death in all age groups that the vaccine has been approved for. Now is the time for communities and families to make the choice to get the updated COVID-19 vaccine.”
In an apparent effort to rebuff widespread concern that Covid shots have resulted in myriad adverse reactions across the nation, ASTHO’s statement asserts that the review of vaccines “includes careful analysis.”
COVID vaccination is most controversial for the young, as CDC data shows that children have almost no risk of death from the virus.
“These processes are robust, have safety at their core, and use the most recent data to evaluate the risks and benefits of vaccinations, including ideal dosage, scheduling, and populations that would benefit most from vaccination,” ASTHO states. “Millions of doses of COVID-19 vaccines have been safely administered worldwide across a wide range of ages and in individuals with underlying health concerns. These efforts have saved countless lives. ASTHO continues to join with other public health and healthcare professionals across the nation to help people learn about vaccine safety and share how COVID-19 vaccines protect the public.”
ASTHO’s endorsement follows the FDA’s March 14 decision to amend the “emergency use authorization” for the Pfizer-BioNTech COVID-19 vaccine, Bivalent, to permit a single booster dose for kids six months to four years old, just two months after their initial vaccinations.
As LifeSiteNews.com pointed out, however, the FDA’s supporting data for child Covid boosters was based off two small-sample studies, which included 24 participants ages 6-23 months, and only 36 participants ages 2-4 years.
Medical freedom attorney and COVID jab critic Aaron Siri called the decision a “new low” for the FDA to base its recommendation on such limited data.
According to the FDA, among the young group, the most common vaccine side effects included “irritability, drowsiness, injection site redness, pain and swelling, decreased appetite, fatigue, and fever,” according to the agency. For the second group, effects included “fatigue, injection site pain, redness and swelling, diarrhea, vomiting, headache, joint pain, and chills.”
ALASKA WATCHMAN DIRECT TO YOUR INBOX
A large swath of Americans, including many Alaskans, remain skeptical about the safety of Covid shots, and the CDC’s own data show that hundreds of thousands of people have suffered serious adverse reactions to the jabs, including death, hospitalizations, strokes and heart attacks.
COVID vaccination is most controversial for the young, as CDC data shows that children have almost no risk of death from the virus. In fact, since January of 2020 to September of this year, children younger than four have comprised 0.1% of all Covid deaths, while those older than 50 make up 83% of Covid deaths.
Next month, thousands of Alaskans are expected to turn out for a high-profile conference organized by Alaska Covid Alliance, a network of doctors, business owners and others who advocate for patients’ rights, early treatment alternatives, and the distribution of vital information that the mainstream media refuses to report on. The group hosted a similar conference in 2021, which drew more than 1,000 attendees and made international headlines.







41 Comments
That is a wicked woman.
YOUR DEAD BABIES CRIES WILL WAKE YOU IN THE NIGHT . ZINK IS A DEMON.
Of course, she does… Just like FAUXXIE… More $$$$$.with no legal ramifications.
When anyone with whom I do not have a loving, personal relationship encourages me to have something injected into my bloodstream, I cry Stranger Danger! and run like the wind.
because the 1st round worked so well! Not happening, im not a lab rat and would rather catch lead posioning 1st.
Very sad the babies don’t have the ability to do the same, their woke parent probably standing in line right now. But hey they so cool!..
SINK ZINK!
Sink the stink Zink!
Don’t forget who her grandfather was … population control freak Al Bartlett. And she “hardly goes a day without thinking of him.”
Dunleavy should now be seen by our fellow Alaskans as not just weak and spineless, this man is evil! He is fully on board with Zink.
I totally agree. Dunleavy has sold us out to the evil of Zink and has never gotten my vote. We have not gotten any of the DNA altering drugs and we would never give them to our children or grandchildren.
Dunleavy is part of the WEF and Zink walks lockstep with her Grandfather.
The apple doesn’t fall far from the treee
Shall I assume you think COVID vaccines are part of some sinister “population control” conspiracy?
You’re catching on, Chuck!
Death by hospital Covid protocol, too. Why do you think they had to work so hard to control the Covid narrative?
No one can force you to get vaccinated, let alone get your child vaccinated. But the fact remains that the overwhelming number of COVID-19 deaths have been among among people who have refused to get vaccinated. Personally I’ve been fully vaccinated and triple-boosted, and I’m getting the new jab this coming Monday. I have no regrets. The one time I DID get COVID, it was a bad sore throat and laryngitis that lasted for about a week. No long-COVID symptoms after that. Take your chances if you like, but if you’d like to see the stories of people who pointedly refused to get vaccinated, only to die horrible deaths while hooked up to ventilators, check out http://www.sorryantivaxxer.com
So your vaccine shots worked, wait you still got covid.
What about all the people who lose there jobs because they did not want the shot. What about all the other people who die because they were not given medication like ivermectin.
You just stay in line like a good little sheep, we will see how your shots are helping you in the future.
Studies show that Ivermectin does not help control COVID symptoms. I’ll take my jab.
my goodness they live….5g is an eye opener for the graphene cult.
Contrary to what Sherri Tenpenny said, metal objects have not started sticking to my skin.
Actually your statement of the overwhelming number of covid-19 deaths have been among the vaccinated is incorrect. There have been many vaccinated people die from covid as well as those who have died and been maimed by the jab. There are also NO datasets supporting the narrative of, “they don’t get as sick.” That’s a pipe dream. Even Pfizer’s own documentation said that their ‘vaccine’ wouldn’t stop transmission or infection. The treatment protocol used in the hospitals is responsible for a million deaths alone. When you do the research you will see that the protocol almost guarantees a negative outcome. But you are either a troll or have drank so much of the kool-aid that you’ll never understand or even want to understand. Sad commentary.
People do not die from covid. They die from other factors. You need to do some serious research. Those who have died have taken the experimental covid vaccine and they are the ones spreading it. Have you not seen reports of the many who just suddenly die. It is your choice to get jabbed. Hope you don’t get the wrong one. Your stats are incorrect as you get them from fake news. Do some research. Check with Americafrontlinedoctors.com. Quit spreading lies. My sister is a nurse–she says ventilators are not meant to use for covid and the drug given at the hospitals ruined kidneys. Those people did not die from covid. Those of us who didn’t take the jab area live and well.
People of all ages die suddenly. It happened before COVID and will continue after.
IF YOU THINK ZINK DOESN’T KNOW ABOUT THE REAL RISKS OF THIS CRAP, GO TO DAILYCLOUT.IO AND READ THE INFORMATION THAT SHOWS THAT THEY ALL KNEW OR IF THEY DIDN’T, THEY DON’T WANT TO KNOW. ALL OF OUR STATE AND FEDERAL ELECTED POS’S ARE GUILTY OF KILLING AND INJURING KIDS ALL OVER THE COUNTRY THROUGH THEIR CORRUPTION, IGNORANCE OR BY DESIGN.
https://dailyclout.io/46-pages-foiaed-from-cdc-leaders-2021-reveal-fauci-collins-white-house-nih-hhs/
100,000 medical professionals say one thing, and a right wing lawyer and a couple wacko doctors disagree. I don’t know which way to turn!
Sometimes, there’s one only person willing to say, “The emperor isn’t wearing any clothes.” I’d rather be that person (or follow him) than one of the fools pretending to admire the emperor’s stunning new outfit.
100,000 “medical professionals?” So, not even necessarily medical doctors…just medical professionals? Please provide your proof for these 100,000 medical professionals.
Conspiracy theories are a dime-a-dozen in the Social Media Age.
When you talk, we know that your words are a theory Chuck. When we talk and give facts, we know that our comments prove that there is a conspiracy!
It is all about control and money. The so called medical professionals made a lot of money pushing the experimental vaccine. Stop watching fake news and do some real research. There is an Alaskan Alliance meeting in Anchorage soon. These doctors and other medical professionals share the truth about what the vaccine has done to people. Not only deaths but terrible physical long range medical problems. Those doctors pushing the jab only get their info from the AMA. They have done no research. Do your own research.
Actually, Mike, you could not be more incorrect. The Great Barrington Declaration was not “a couple of right wing doctors” it was over 60,000 medical professionals, nurses and physicians and scientists, ALL signing on to a statement that COVID was being handled TOTALLY inappropriately, actual science and scientific discussion was being ignored to our collective detriment, and that alternative treatments WERE shown to be effective and should not be summarily descried as ineffective in order to justify the EUA. Many of the signers of the GBD were nobel laureates, and scientists at the absolute pinnacle of their careers, who’s career, credential and livelihood were destroyed by not letting bureaucrats have the final say on what is, and is not, evidence and history.
Let me help you Mike, your the type that most definitely needs the shot and all the boosters!
I got the shots and all the boosters, and I’ll be getting the update booster tomorrow. No regrets.
Governor Dunleavy, you made a seriously grave mistake in backing and recommending genocidal Dr. Zink. Humanity is being destroyed because of her and her evil partners in crime.
FOH precisely! Which is why dunleavy and zinc need to be arrested for crimes against humanity!
AMEN DAVE…genocide has consequences…
Off-topic, but need to share! “FEMA wis [sic-should be “is”] scheduled to implement an emergency broadcast alert takeover on either October 4th or October 11th at 2:22 PM EASTERN, and intel suggests that the frequency used may trigger graphene in the both the vaxxed and unvaxxed.” SGT Report: RED ALERT WARNING: FEMA ZOMBIE 5G APOCALYPSE — CALLENDER | VLIET
https://rumble.com/v3kgmyy-red-alert-warning-fema-zombie-5g-apocalypse-callender-vliet.html
OCT 4TH, FEMA, DARPA, DEPOPULATION AGENDA
https://www.brighteon.com/02eeb8d9-5276-4553-b774-8329e8509f9b
…ANYONE recommending or giving these bio-jabs need to be criminally charged with crimes against humanity!!
Nuremberg code
So who or what starts the revolution? It’s time!
Protect your babies!! Say NO!!!
‘Tucker Carlson – ‘This is A Spiritual Battle’ – Goes Scorched-Earth Against ‘Anti-Human’ New World Order’ 9/30/23
https://www.brighteon.com/3cb8f01c-1c74-4be7-be0c-f86110341023