
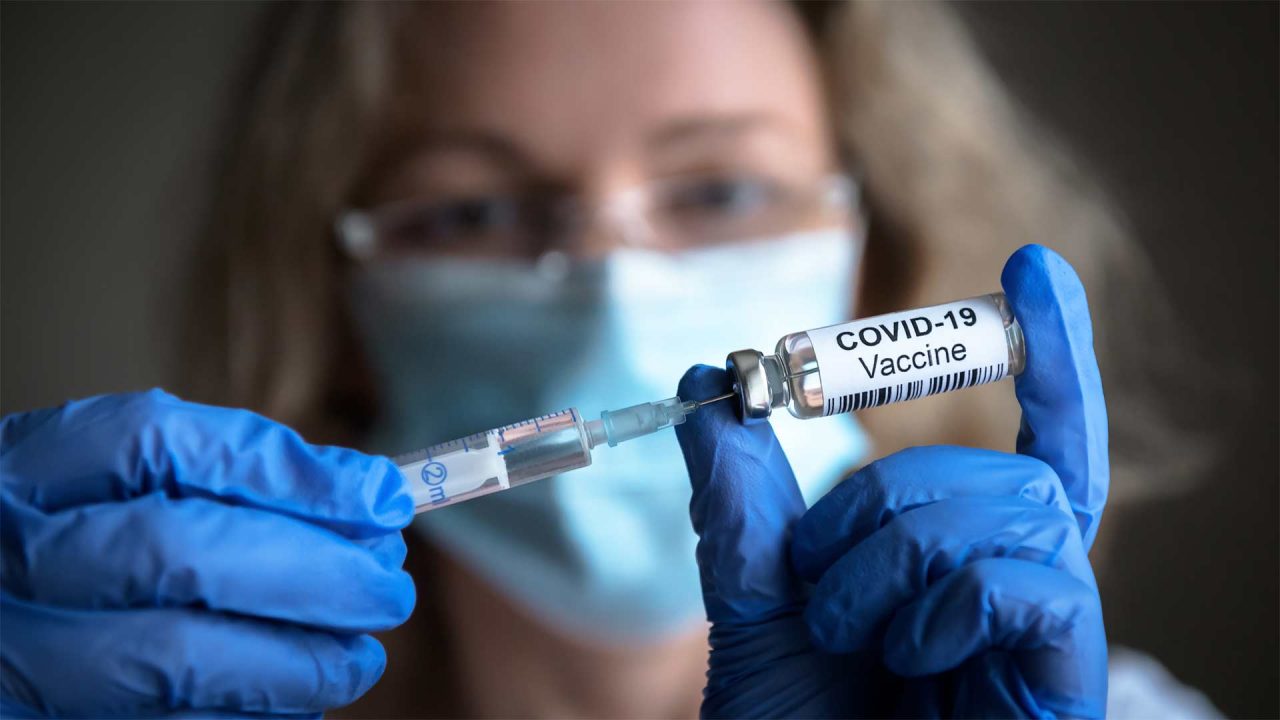
Due to reports of dangerous blood clotting in the brain associated with the Johnson & Johnson COVID vaccine, the CDC has recommended an immediate pause on using the experimental shot which, like all other COVID vaccines, has only been approved for emergency use.
As of April 12, more than 6.8 million doses of the J&J shot have been administered across the nation, including more than 11,000 in Alaska. The CDC and FDA announced on April 13 that they are now investigating six reported U.S. cases of a “rare and severe type of blood clot in individuals after receiving the J&J vaccine.” All six reported cases occurred in women between the ages of 18 and 48 with symptoms occurring six to 13 days after the shot was administered. The New York Times has reported that one of the women has died and a second has been hospitalized in critical condition in Nebraska.
The CDC is analyzing the six cases and their potential significance.
“Until that process is complete, we are recommending a pause in the use of this vaccine out of an abundance of caution,” the CDC stated on April 13. “This is important, in part, to ensure that the health care provider community is aware of the potential for these adverse events and can plan for proper recognition and management due to the unique treatment required with this type of blood clot.”
ALASKA WATCHMAN DIRECT TO YOUR INBOX
The CDC warns that those who have received the J&J vaccine, and later develop “severe headache, abdominal pain, leg pain, or shortness of breath within three weeks after vaccination should contact their health care provider.”
The Alaska Department of Health and Social Services hosted a media briefing April 13 to address the CDC warning.
As of April 12, there have been 11,178 Johnson & Johnson vaccine doses administered in Alaska out of 35,500 doses allocated to the state. Alaska has had nine reports of adverse reactions to the J&J shot since April 2.
State Epidemiologist Joe McLaughlin repeatedly underscored that the blood clotting associated with the J&J appear to be very rare, and that all six of the identified cases are from the Lower-48. He added that those who did receive the J&J shot should be aware of adverse reactions and report them.
McLaughlin noted that the pause on the J&J shot could last a few days or weeks, or even longer.
“This will certainly increase the level of concern among some people,” he said, adding that the blood clotting concerns are related specifically with the J&J vaccine and not with Pfizer or Moderna.







11 Comments
The whole plandemic has been a culling of the population, Those in Govt that urge these poison injections know it is a murderous scheme of the demonic globalists and they play their role, The Influenza that usually plagues our nation is gone yet these complete dolts that are in our legislature will not say a word to warn the people they were elected to represent. They are a part of the culling…..They are the nazi guards pushing the people into the ovens….. When will you wake up? The Watchman must warn.
John
I totally agree with you 100%! Who will stand for truth in the legislature and protect the uninformed of the risks involved with the vaccine the senseless wearing of the masks?? Our Govt is planning to depopulate us, using the ignorance and trust of the people as a guise! Yes, Watchman please warn them all!
What an untested vaccine.
Right, it’s crazy to take any of these vaccines
Don’t get any shot
The Georgia Guidestones message is that these sick demon elite that run this world Klaus Shwab and Bill Gates of Hell demand a world population of 500 million people. To achieve that there must be a reduction of 6.5 billion souls . Yes Genocidal murder… Chem sprays and Vaccines are funded by the Gates foundation, The Johnson and Johnson Vaxx is not the only vaxx causing death ,,, They all are. it is a culling.
He’s done it before, look up Gate’s history of vaccinations in India and attempting to do the same in Nigeria (as I remember, it was a country in Africa) that refused his vaccine. You will be sickened by this murderer of future populations.
https://www.facebook.com/photo/?fbid=10164912432550646&set=a.98075020645&__cft__%5B0%5D=AZVUekXM9x2nXtGxSU-TzrOhOmmJCMVTIEi-7jmI2qK_xz4j-q4H1yEA5kNpOfutss089lHgpUIlxyHTewBuMUjs1RkOMyrdo5wKPi3ygVB09DmP9iZO8Jworx66KMo-0Njp9lJribx_xR1lgYBv1LaWRIFgDgOZ2_EbPGq_cv5O364jFjQ5VSwJuH9lbJDHMAo&__tn__=EH-y-R
Thank you for keeping us updated
How interesting that the J & J vaccine, which is actually a true vaccine and less “experimental” than the other, is now “ on pause”. …because 6 out 6.8 MILLION had a rare reaction. Seriously?? I don’t want ANY shot.. but that one seemed the lesser of evils. How many bad reactions did the other have?? Hundreds? Thousands? Seems fairly suspect.
Agree. That is the shot I might consider getting for upcoming travel. Good grief.
Just to keep it in perspective, the Astra Genica shot has just as many blood clots percentage wise yet has been deemed far safer than not being vaccinated. Why J&J? It’s not the mRNA? It’s not influenced by Bill Gates et al? Very curious. As mentioned it actually is the only one to use traditional immune boosting technology through the use of neutralized virus.
If communists er- democrats had it their way, they would have never stopped. I’m waiting for “vaccination check points” on the Glen and Seward highways. Either get the jab or turn around. It’s because that’s how communists roll. Getting closer to Reset Day..